9.7 Linear structures: strands, cords, rhizomorphs and stipes
Formation of parallel aggregates of morphologically similar hyphae is common among Ascomycota and Basidiomycota. The most loosely organised, mycelial strands and cords, can provide the main translocation routes of the mycelium, and so develop in circumstances that require large scale movement of nutrients (including water) to and from particular sites. Mycelial strands originate when young branches adhere to older leading hyphae and weave them together (Fig. 13). Further localised growth and incorporation of other hyphae it may meet leads to increase in size of the strand.
Anastomosis between the hyphae of the strands consolidates them and narrow hyphal branches (tendril hyphae) from the older regions of the main hyphae intertwine around the other hyphae (Fig. 13). From the beginning, some of the central hyphae may be wide-diameter, thin walled so-called vessel hyphae and in older strands narrow, but thick-walled, fibre hyphae appear, running longitudinally through the mature strands. Strand formation occurs in ageing mycelium on an exhausted substrate when the hyphae are likely to be the main repositories of nutrients (especially nitrogen) and it has been argued that stranding results from the limitation of new growth to the immediate vicinity of the remaining nutrient. As long as the strand is the main supplier of nutrient the integrity of the strand will be reinforced, but when the strand encounters an external source greater than its own endogenous supply the stimulus to cohesive growth is lost and spreading, invasive, hyphal growth envelops the new substrate.
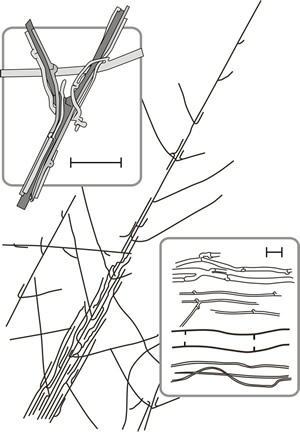 |
Fig. 13. Drawing of hyphal strands of Serpula lacrymans. Strands originate when branches of a leading hypha form at an acute angle to grow parallel to the parent hypha which also tends to grow alongside other hyphae it may encounter. Anastomoses between the hyphae of the strands consolidates them and narrow hyphal branches (‘tendril’ hyphae) emerging from the older regions grow around the main hyphae and weave them together. The strand is shown in a general habit sketch running diagonally across the figure and the top panel shows tendril hyphae intertwined around main hyphae. The bottom panel shows some of the cell types encountered in strands, with undifferentiated hyphae at the top then tendril hyphae, a vessel hypha and fibre hyphae, scale bars = 20 μm. Modified from Moore, 1995. |
Although mycelial strands contain morphologically differentiated hyphae (Fig. 13), their constituent hyphae are relatively loosely aggregated. Certain fungi produce highly differentiated aggregations of hyphae with well developed tissues (Fig. 14). These structures are very root-like in appearance and are called rhizomorphs. Rhizomorphs differ from strands by having extreme apical dominance with a highly organised apical growing point. The apical region of the rhizomorph contains a compact growing point of tightly packed cells, protected by a cap of intertwined hyphae in (and producing) a mucilaginous matrix. Behind is a medullary zone containing vessel hyphae composed of swollen, vacuolated and often multinucleate cells surrounded by copious air- or mucilage-filled spaces. The medullary region forms a central channel through the rhizomorph and, in mature tissues, is traversed by narrow-diameter, thick-walled fibre hyphae (Fig. 14). Towards the periphery of the rhizomorph, the cells are smaller, darker, and thicker-walled, and there is a fringing mycelium extending outwards between the outer layers of the rhizomorph, resembling the root-hair zone of a plant root.
The similarity, at least in microscope sections, with the plant root has prompted the suggestion in older literature that rhizomorph extension results from meristematic activity. However, a meristem-like structure would be totally alien to the growth strategy of the fungal hypha; meristems do not occur in fungi. The impression of central apical initials giving rise to axially arranged tissues is undoubtedly an artefact caused by sectioning compact aggregations of parallel hyphae. Ultrastructural, especially scanning electron microscope, observations clearly reveal the hyphal structure of the rhizomorph tip, but the appearance of the rhizomorph body being made up of parallel bundles of hyphae must mean that increase in rhizomorph diameter is associated with highly regulated hyphal branching.
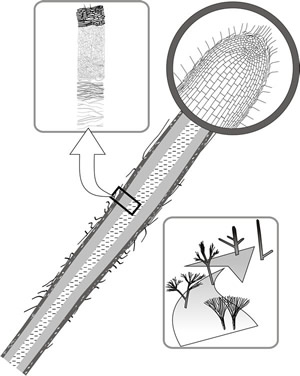 |
Fig. 14. Rhizomorph structure. The diagonal diagram is a sectional drawing showing general structure, with the apical region magnified to show the appearance of a growing point of tightly packed cells. Behind the tip is a medullary zone containing swollen, vacuolated and often multinucleate cells surrounded by copious air- or mucilage-filled spaces. The medullary region forms a central channel through the rhizomorph and, in mature tissues, is traversed by narrow fibre hyphae and wide-diameter vessel hyphae, the microscopic appearance being indicated in the drawing in the top left panel. The panel at bottom right depicts mycelial fans, strands, cords and rhizomorphs as a series showing increasing apical dominance (Rayner et al., 1985). Modified from Moore, 1995. |
Usually, rhizomorphs are initiated as compact masses of aggregated cells the ultimate origin being ascribed to locally enhanced acute-angled branching of some marginal hyphae in a mycelium; a phenomenon described as ‘point-growth’; the linear organs originating from originally unpolarised hyphal aggregations which become apically polarised. Mycelial strands and rhizomorphs are extremes in a range of hyphal linear aggregations that are related together in a hierarchy depending on increasing apical dominance (Fig. 14). Their essential function is the translocation of nutrients, but they also penetrate the substratum, explore and migrate.
For example, strands form when there is a need to channel
nutrients towards developing fruit bodies, and they are also formed by
mycorrhizal fungi to radiate into the soil, where they greatly supplement the
host plant’s root system and gather nutrients for the host. In saprotrophic
phases, strands are also migratory organs, extending from an
existing food base to explore nutrient-poor surroundings for new nutrient
sources. Strands of Serpula lacrimans, the dry-rot fungus, are able to
penetrate several metres of brick-work from a food base in decaying wood and to
overgrow plastic and many inert building materials. The strands hasten capture
of new substrate by increasing the inoculum potential of the fungus at the point
of contact with it, aiding capture of resources and providing translocation
routes in both directions. The distribution of strands around a food base
changes with time as hyphae are resorbed; that is, digested from the inside to
regain nutrient components. Redistribution of the nutrients recovered from old
strands enable migration of the colony from place to place.
The prime example of rhizomorphs is usually Armillaria mellea, a
pathogen of trees and shrubs, which spreads from one root system to another by
means of its ‘boot-lace’ rhizomorphs. Here, again, the structure serves
translocatory and migratory functions and, as with strands, translocation is
bidirectional, glucose being translocated towards and away from the apex
simultaneously. In moist tropical forests aerial rhizomorphs, mainly of
Marasmius spp., form a network which intercepts and traps freshly fallen
leaves, forming a suspended litter layer. In describing the
structure of litter‑trapping rhizomorph networks in moist tropical forests,
Hedger et al (1993) showed that the rhizomorphs have a reduced fruit
body cap at their tips, so the stems (= stipes) of fruit bodies should also be
included in this discussion. These two linear organs are functionally very
similar, the fruit body stipe translocates nutrients to the fruit body cap, and
as many fruit bodies are served by radiating strands which convey nutrients
towards the fruiting structure the junction between strand and stipe can be
obscure. The term ‘radicating’ is used to describe fruit bodies whose stipes are
elongated into root-like pseudorhizas which extend to the soil
surface from some buried substrate. Even in species which do not normally
produce pseudorhizas, they can be induced by keeping fruiting cultures in
darkness (Buller, 1924) whereupon the stipe base can extend for many centimetres, driving
the fruit body primordium on its tip towards any source of light. Rhizomorphs?
Pseudorhizas? Extending stipes? What they are called is less important than the
implication that a close morphogenetic relationship underlies all fungal linear
hyphal aggregations.
In this Section and the next, the fungal structures are described in terms of the apparent role of the structures in their ecosystems. These attributes, or ‘functional traits’, are the tools the organism uses for survival, reproduction, and dispersal. Fungal traits have been used to establish identification and taxonomy but rarely for gaining insight into fungal ecology (Halbwachs & Bässler, 2021). In contrast, plant scientists have used functional traits for many years to understand plant ecology. Think, for example, of how flower colour attracts insects, and nectaries reward those insects that (inadvertently) collect the flower’s pollen and then pollinate other blossoms. Halbwachs & Bässler (2021) demonstrate how trait-based functions can help us to understand fungal ecological behaviour but conclude that many interpretations are based on plausibility, rather than evidence, so much further research needs to be done.
Updated June, 2021
