7.2 Formation of heterokaryons
Hyphal anastomosis, of course, is the fusion between hyphae or hyphal branches, and we have described the process of breakdown of two hyphal walls and union between two separate plasma membranes to bring the cytoplasms of the fusing hyphae into continuity with each other. It is the consequence of this that’s important here: once they are in continuity, they can exchange nuclei and other organelles. Of course, this happens in the gametic fertilisation which is a prelude to sexual reproduction in animals and plants.
Fungi differ because anastomosis is not limited to sexual reproduction; rather, hyphal fusions are essential to the efficient functioning of the mycelium of filamentous Ascomycota and Basidiomycota. Anastomoses convert the initially radiating system of hyphae into a fully interconnected (and three-dimensional) network. Hyphal fusions are common within the individual mycelium as it matures. The interconnections they establish enable transport of nutrient and signalling molecules anywhere in the colony.
| Table 1. Frequency of the occurrence of heterokaryons amongst fungi isolated from nature | ||
| Species | Number of isolates |
% of isolates found to be
heterokaryons |
| Aspergillus glaucus | 15 |
13 |
| Penicillium cyclopium | 16 |
25 to 50 |
| Sclerotinia trifoliorum | 10 |
60 |
Heterokaryons do occur in nature (Table 1). Evidently, higher fungi are fully equipped with the machinery necessary for hyphal tips to target other hyphae and this is part of normal mycelial development; it is not a specialisation of the sexual cycle, nor a peculiarity of in vitro cultivation. It is complex machinery, though, because during normal mycelial growth vegetative hyphae usually avoid each other (known as negative autotropism), behaviour that promotes exploration and exploitation of the available substrate. Anastomosis requires that hyphae grow towards each other (called positive autotropism). How and why the usual avoidance reactions between hyphae become reversed is unknown but, evidently, it is a change in the behaviour of hyphal tips that occurs as the mycelium matures (and may depend on local population density).
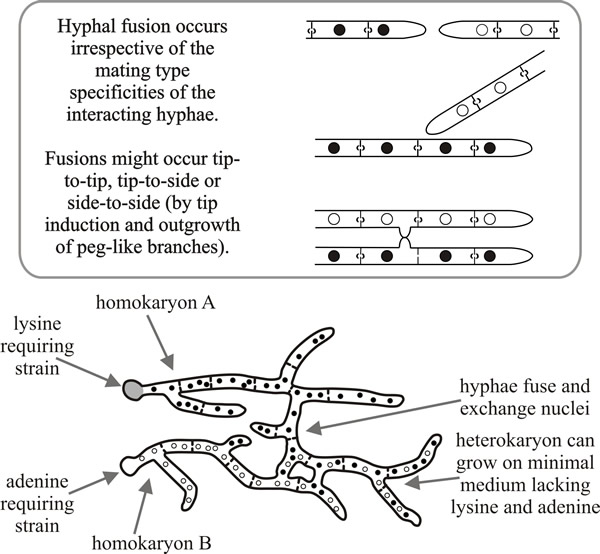
Higher fungi, therefore, have a mechanism that promotes cell fusions to an extent that is never encountered in animals, plants, or even lower fungi and their more primitive relatives. The process brings two different cytoplasms into one (joint) mycelium, and also brings nuclei originating from two different mycelia into the same (joint) cytoplasm (Fig. 2). The genetic consequences of this for the heterokaryon are important. Alleles in separate nuclei in a heterokaryon will complement each other; in the example shown in Fig. 1 a heterokaryon formed from a lysine-requiring strain (a nutritionally-deficient ‘auxotroph’) and an adenine auxotroph will be able to grow on a minimal medium (one that contains neither of these nutrients), although the two constituent homokaryons will not.
The ratio of different nuclei in a heterokaryotic mycelium may influence both its morphology (branching frequency, for example) and growth rate (by affecting hyphal extension). Thus, the phenotype of a heterokaryon is determined by the balance of nuclei in the heterokaryon. Equally, the ratio of different nuclei will itself be influenced by the environmental and growth conditions.
 |
|
| Fig. 2. Homokaryosis and heterokaryosis as illustrated by Aspergillus nidulans (Ascomycota). Top, diagrams of the different ways in which hyphal fusion may occur between homokaryons. Bottom, diagram of a heterokaryon being formed by two homokaryons. When the nuclei in the heterokaryotic mycelium are of two or more genotypes, alleles in separate nuclei in a heterokaryon may complement each other. In this diagrammatic example a heterokaryon formed from a lysine auxotroph and an adenine auxotroph will be able to grow on minimal medium, although the two constituent homokaryons will not. |
| Table 2. Effect of environmental conditions on the nuclear ratio in a heterokaryotic mycelium of Penicillium cyclopium (A and B type nuclei). | ||
| Growth medium | % of the two nuclei in the
heterokaryon |
|
A type |
B type |
|
| Minimal | 9 |
91 |
| Complex (apple pulp) | 52 |
48 |
Table 2 shows the effect of environmental conditions (in terms of the nature of the medium) on the nuclear ratio in a heterokaryotic mycelium of Penicillium cyclopium (a fungus that commonly causes fruit spoilage with production of toxins), where the B type nuclei predominated on a minimal medium, but both nuclei were equally represented in heterokaryons grown on an apple pulp medium. Thus, the ratio of nuclear types in a heterokaryon can vary within wide limits. In this case, the interpretation is that the complex medium provides a selection pressure favouring maintenance of the heterokaryon, the minimal medium does not, and the B type nucleus has some selective advantage. Note the contrast with the example shown in Fig. 2C, where the minimal medium favours the heterokaryon comprising ade- and lys- nuclei. In another example of experiments with heterokaryons of Neurospora crassa formed between colonial mutants (with a highly branched mycelium that expands slowly) and non-colonial strains (with a sparsely branched mycelium that expands rapidly), the heterokaryons exhibited the non-colonial phenotype when nuclei of the two mutants are present in a ratio of 1:1, but a colonial phenotype when the heterokaryon contained more colonial than non-colonial nuclei. Thus, the ratio of nuclei present in a heterokaryon also influences its morphological phenotype, that is, the branching frequency and rate of hyphal extension.
Evidently, there are several consequences of heterokaryosis (Strom & Bushley, 2016):
- different nuclei provide genetic variation in the mycelium;
- phenotype of the mycelium depends on interaction of all nuclear types;
- phenotype of the mycelium can vary in different areas of the same mycelium, depending on the localised nuclear ratio;
- the heterokaryon has the physiological flexibility to respond to different nutritional conditions;
- heterokaryons increase genetic diversity independently of sexual reproduction;
- provides a route for cytoplasmic inheritance (mitochondria, viruses, plasmids, prions, see below, Section 7.9).
Updated July, 2019
